If DNA is known to be the support of the genetic information, it is also a polymer carrying a series of phosphate groups. In contact with water molecules, phosphate groups hydrate and ionise in a such way that the DNA chain may be considered as a long chain of negatively charged monomers (i.e. a poly-anion). The same number of positive charges has to counterbalance the DNA charges. In vivo the positive charges may be simple ions (potassium, sodium, calcium,…) and/or belong to other cell macromolecular components (proteins,..). The team is interested in DNA as a polyelectrolyte, working on model systems in vitro. According to the valency of counterions, various phenomena can be observed. In the presence of monovalent cations, the interactions between DNA molecules in solution are repulsive. When the valency of the cations is higher or equal to three, or in presence of basic proteins, an attraction between DNA molecules gives rise to a phase separation i.e. to the formation of a phase very concentrated in DNA coexisting with a dilute DNA solution. According to the length ans concentration of DNA, molecules collapse and/or aggregate.
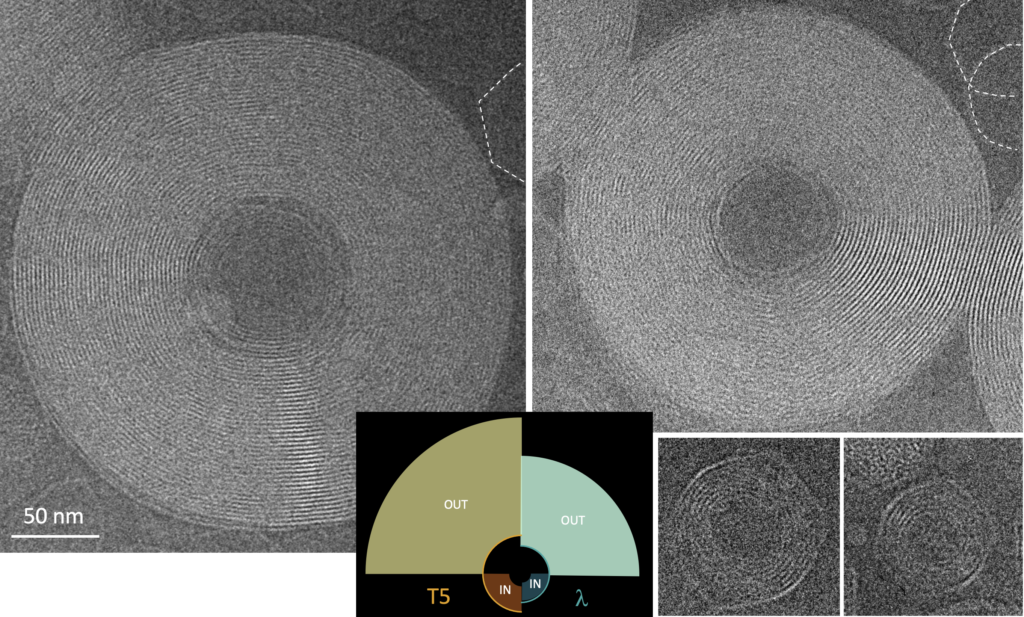
DNA toroids
DNA toroids are complex liquid crystalline objects that combine properties of both biological and material sciences objects. They spontaneously form in vitro upon DNA condensation: a DNA chain in good solvent adopts an extended coil conformation due to the electrostatic repulsions between its negatively charged segments and its bending rigidity; in the presence of multivalent cations of basic proteins, it condenses into compact toroidal structures. This behaviour is a general phenomenon, typical of semi-flexible polyelectrolytes, and is also found in a variety of synthetic or biological molecules, for example actin or collagen.
These amazing objects have intrigued biophysicists and physicists, both experimentalists and theoreticians since their discovery in 1976. They are considered as model objects for understanding high density packing of DNA in many biological systems (viruses, sperm cells, bacterial nucleoids). DNA toroids also inspire gene delivery and nanodevice design.
We have designed an experimental procedure to prepare DNA toroids in nano- and meso- sizes, to control a wide range of DNA curvature. Using cryo-EM and cryo-ET, we explore their 3D architecture and local order between DNA helices, and collaborate with theoretical physicists.
DNA bundles
In the presence of multivalent cations or basic proteins, short-length DNA fragments (typically 50-500 nm) aggregate into bundles. Among condensing agents, protamines are oligo peptides with high charge density, responsible for sperm chromatin condensation. We explore the mechanisms involved in the formation of soluble protamine-complexes using a combination of experimental and simulation approaches. More generally, this should help re-interpreting puzzling behaviors reported for a large class of strongly charged polyelectrolyte systems.

Related publications
- Leforestier, A., & Livolant, F. (2009). Structure of toroidal DNA collapsed inside the phage capsid. Proceedings of the National Academy of Sciences, 106(23), 9157-9162. doi.org/10.1073/pnas.0901240106
- Leforestier, A., Šiber, A., Livolant, F., & Podgornik, R. (2011). Protein-DNA interactions determine the shapes of DNA toroids condensed in virus capsids. Biophysical journal, 100(9), 2209-2216. doi.org/10.1016/j.bpj.2011.03.012
- Lansac, Y., Degrouard, J., Renouard, M., Toma, A. C., Livolant, F., & Raspaud, E. (2016). A route to self-assemble suspended DNA nano-complexes. Scientific reports, 6(1), 1-12. doi.org/10.1038/srep21995
- Sung, B., Leforestier, A., & Livolant, F. (2016). Coexistence of coil and globule domains within a single confined DNA chain. Nucleic acids research, 44(3), 1421-1427. doi.org/10.1093/nar/gkv1494
- Barberi, L., Livolant, F., Leforestier, A., & Lenz, M. (2021). Local structure of DNA toroids reveals curvature-dependent intermolecular forces. Nucleic acids research, 49(7), 3709-3718. doi.org/10.1093/nar/gkab197
- Mukherjee, A., de Izarra, A., Degrouard, J., Olive, E., Maiti, P. K., Jang, Y. H., & Lansac, Y. (2021). Protamine-Controlled Reversible DNA Packaging: A Molecular Glue. ACS nano, 15(8), 13094-13104. doi.org/10.1021/acsnano.1c02337
Contact
Amélie Leforestier: amelie.leforestier@universite-paris-saclay.fr
Yves Lansac: yves.lansac@univ-tours.fr